Using a Chemical Time and Temperature Label
There are many ways to measure temperature change and for recording how long a temperature-controlled substance, material, or chemical compound, is maintained outside of its normal required temperature limits for stability, or future use. Time Out is not a new term, or monitoring application, but it is a complex one and certainly needed. The most common is for Pre-Preg and Carbon Fiber materials that must be maintained within strict temperature guidelines for flexibility and strength requirements, plus many Chemical compounds and liquid API’s (Active Pharmaceutical Ingredients). These strict limits also apply to certain bio-logics and vaccines that can only be “out-of-environment” for short periods of time whilst they are being worked with, formulated, or packaged.
These tracking solutions range from RFID tracking and monitoring with expensive tags and readers to complex solution software applications that read and record these expensive electronic devices, attached to the item being monitored, as these pass in and out of the safe temperature zones.
On the low-end monitoring is done by timers with alarms and some form of temperature recorder to show the temperature limits that the materials were exposed to. Combine both elements and you get a Time and Temperature limit that the materials were within showing how long this was for.
But in some applications, simplicity may be a better solution like – a time and temperature breach indicator label, instead of reading separate items and manually recording these for record purposes.
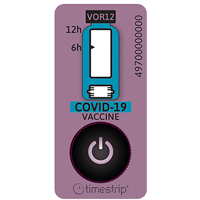
Timestrip Plus labels and Run Timer Labels are used for many applications ranging from monitoring event times related to an expiration of effect use life to monitoring vaccine and bio’ shipments as these pass through a logistics operation, or down the cold chain supply chain. However, if you take a Timestrip Plus label that contains a time element and a temperature limit indicator for an excursion, or the work environment temperature, you have a truly unique and simple way of measuring TOTCE!
The advantage that these labels offer is quite simple by the fact that once the label is outside of its “strike” temperature the chemical will flow down the time indicator scale to show how long the label is outside of the lower product stability-controlled temperature environment. When the material or chemical substance with the label attached is returned to the temperature-controlled environment the chemical will stop flowing and only now show the “Out-Time”. If this item is taken in and out many times the label will begin to form a total aggregated TOTCE as the chemical flows and stops each time it moves in and out of the two environments.
A record of each individual “Out-Time” can be captured by taking a photo with a Smart Phone which will show the time and date that it was taken, and this can be stored as a permanent record linked to the serial number on the label itself that will be clearly seen in the photograph.
Sometimes simple is the best approach based upon the old adage of the “KISS” philosophy, so contact Timestrip today if you want to find out more about this TOTCE solution offering.